In order to patricipate in the following studies, individuals must be willing to give written informed consent. and be able to comply with study regulations and visits. Compensation may be provided for participation *
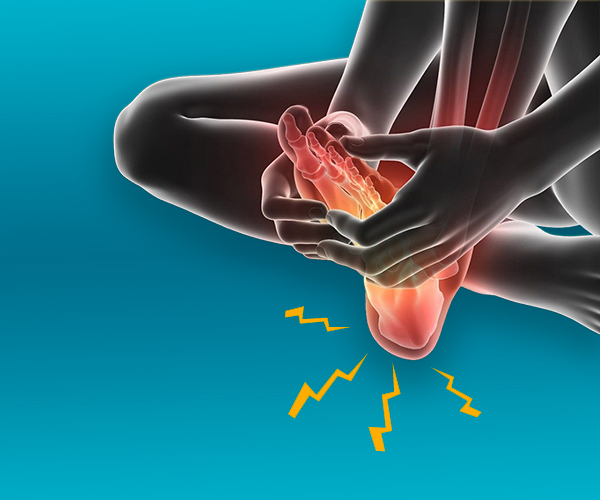
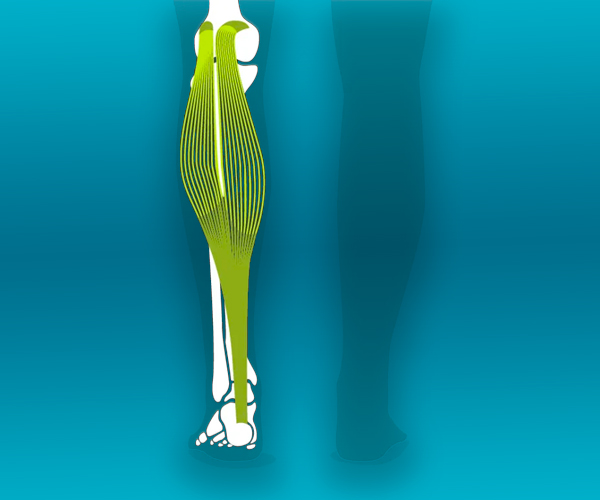
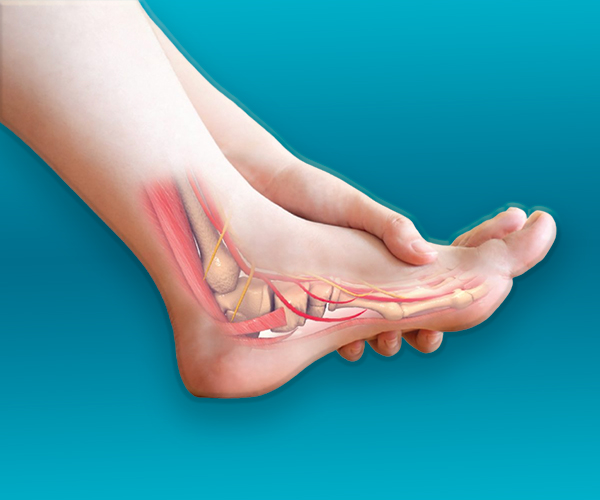
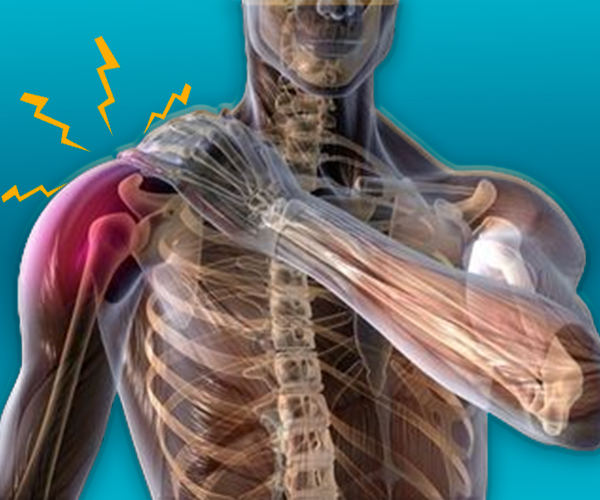
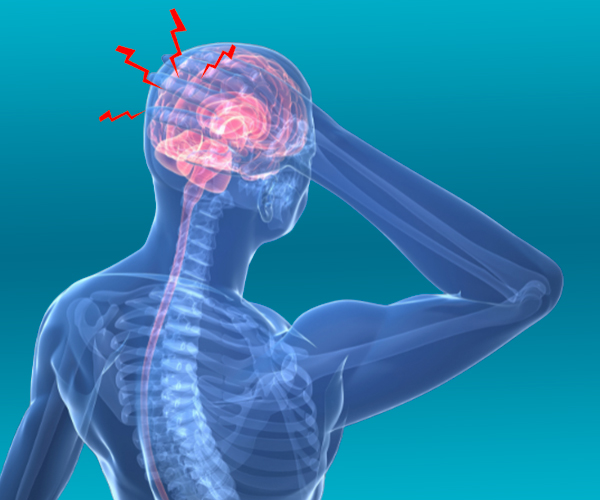
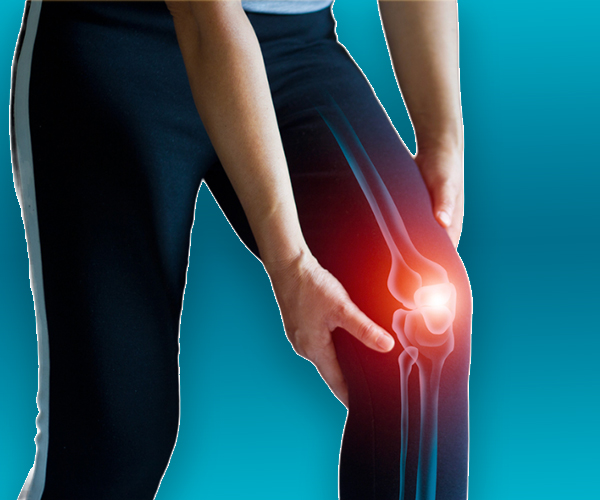
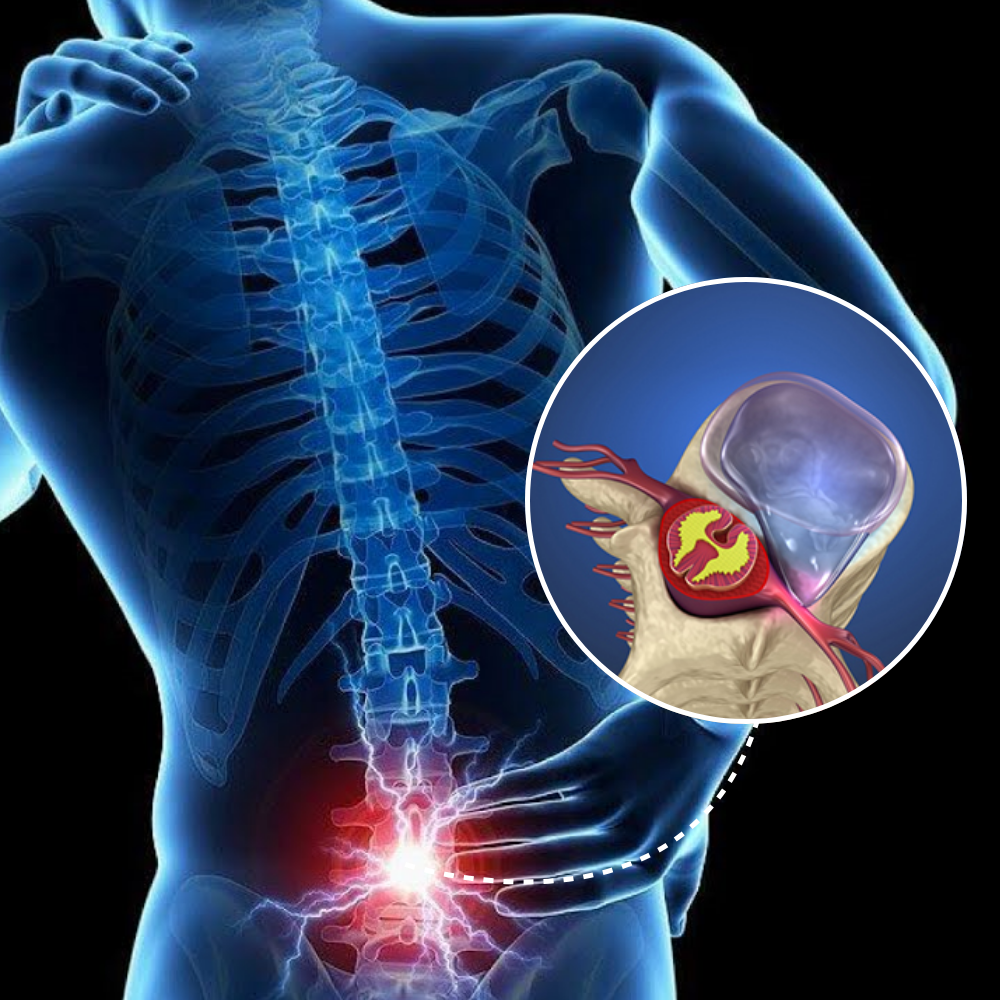
We love our patients, so feel free to ring during normal business hours and ask about current trials.
Overland Park, Kansas - (Kansas City area)
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo. Aenean condimentum lectus tellus, a vulputate enim malesuada id. Nunc in lacus lectus. Pellentesque sit amet malesuada tellus.
Orci varius natoque penatibus et magnis dis parturient montes, nascetur ridiculus mus. Maecenas quis dignissim turpis. Nunc at suscipit ipsum, a ultrices tellus. Duis a nulla vestibulum, facilisis massa at.
Choose the insurance pellentesque lectus nisi, varius eu tincidunt sit amet, feugiat sed ligula. Nulla mollis odio vitae mattis lobortis.
Choose the insurance pellentesque lectus nisi, varius eu tincidunt sit amet, feugiat sed ligula. Nulla mollis odio vitae mattis lobortis.
Choose the insurance pellentesque lectus nisi, varius eu tincidunt sit amet, feugiat sed ligula. Nulla mollis odio vitae mattis lobortis.
10995 Quivira Road, Overland Park,
Kansas 66210, United States