Primary objective:
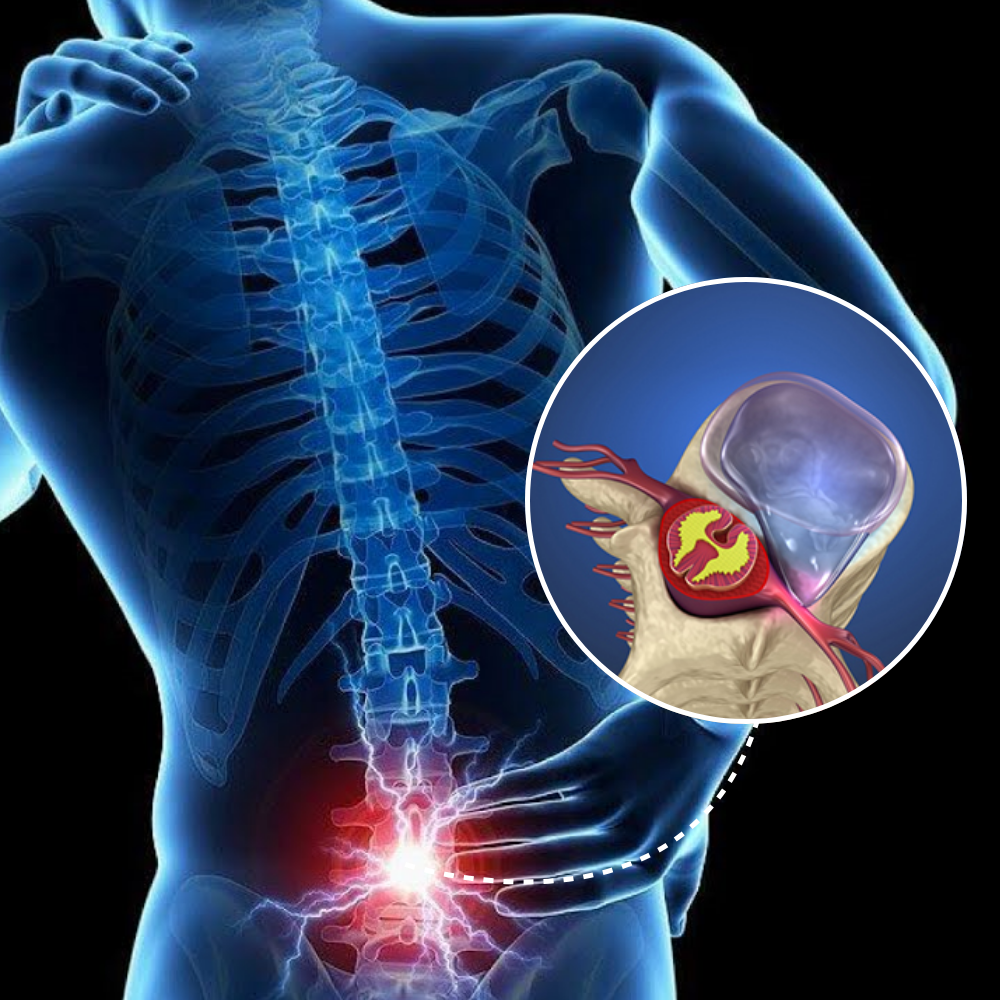
A Phase 3 Randomized Clinical Trial Investigating the Efficacy and Safety of a Lumbar Injection in the Treatment of Pain associated with Back and Leg pain.
Inclusion Criteria:
Exclusion Criteria:
We love our patients, so feel free to call during normal business hours and ask about current trials.
Overland Park, Kansas - (Kansas City area)
10995 Quivira Road, Overland Park,
Kansas 66210, United States